Preço normal
¥0.00 CNY
Preço normal
Preço de saldo
¥0.00 CNY
1. Synonyms
GUM XANTHAN;GLUCOMANNAN MAYO;GALACTOMANNANE;XANTHANGUM,FCC;XANTHANGUM,NF;XANTHATEGUM;Xanthan Gummi;XANTHAN NF, USP
| Xanthan gum Chemical Properties |
| Melting point | 64.43 °C |
| storage temp. | Hygroscopic, -20°C Freezer, Under inert atmosphere |
| solubility | Soluble in water giving a highly viscous solution, practically insoluble in organic solvents. |
| form | Solid |
| color | Off-White to Pale Yellow |
| Odor | sl. organic odor, tasteless |
| Merck | 14,10057 |
| Stability: | Stable. Combustible. Incompatible with strong oxidizing agents. |
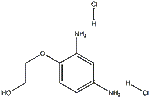
| InChI | InChI=1S/C8H12N2O2.2ClH/c9-6-1-2-8(7(10)5-6)12-4-3-11;;/h1-2,5,11H,3-4,9-10H2;2*1H |
| InChIKey | VXYWXJXCQSDNHX-UHFFFAOYSA-N |
| SMILES | C(O)COC1=CC=C(N)C=C1N.[H]Cl.[H]Cl |
| LogP | 3.926 (est) |
| CAS DataBase Reference | 11138-66-2(CAS DataBase Reference) |
| EPA Substance Registry System | Xanthan gum (11138-66-2) |
| Xanthan gum Usage And Synthesis |
| Description | Xanthan Gum is a long chain polysaccharide, which is made by mixing fermented sugars (glucose, mannose, and glucuronic acid) with a certain kind of bacteria. It is mainly used to thicken and stabilize emulsions, foams, and suspensions. Xanthan gum is widely used as a food additive to control the rheological properties of a wide range of food products. In manufacturing, xanthan gum is used as a thickening and stabilizing agent in toothpastes and medicines. It is used to make medicine for lowering blood sugar and total cholesterol in people with diabetes. It is used as a laxative. Xanthan gum is sometimes used as a saliva substitute in people with dry mouth (Sjogren's syndrome). |
| Occurrence, Isolation | Xanthan gum, the extracellular polysaccharide from Xanthomonas campestris and some related microorganisms, is produced on a nutritive medium containing glucose, NH4Cl, a mixture of amino acids, and minerals. The polysaccharide is recovered from the medium by isopropanol precipitation in the presence of KCl. |
| analysis of bacterial polysaccharide | Xanthan gum is a high-molecular-weight polysaccharide gum produced by a pureculture fermentation of a carbohydrate with the gram-negative bacteria Xanthomonas campestris. This natural polysaccharide is widely used in the food industry and to a lesser extent in the pharmaceutical industry. Xanthan gum is monographed in the USP28/NF and in the PhEur. It is soluble in hot and cold water, as well as being stable under acidic and alkaline conditions (pH 5–13). The primary structure of xanthan gum contains D-glucose and D-mannose as the dominant hexose units, along with D-glucuronic acid. The trisaccharide side chain consists of two D-mannose residues and one D-glucuronic acid residue occurring as mixed K+, Na+, and Ca++ salts. Through the association of xanthan molecules, it is thought that a quaternary structure arises through the charged trisaccharide side chains. Xanthan gum is a water-soluble polymer. Neither USP nor PhEur provides an HPLC method for determination of identity or purity. |
| References | [1] http://www.webmd.com [2] https://en.wikipedia.org/wiki/Xanthan_gum |
| Chemical Properties | viscosity of 1% solution 1,200-1,600 mPas |
| Chemical Properties | Xanthan gum occurs as a cream- or white-colored, odorless, freeflowing, fine powder. |
| Uses | Xanthan Gum is a gum obtained by microbial fermentation from the xanthomonas campestris organism. it is very stable to viscosity change over varying temperatures, ph, and salt concentrations. it is also very pseudoplastic which results in a decrease in viscosity with increasing shear. it reacts synergistically with guar gum and tara gum to provide an increase in viscosity and with carob gum to provide an increase in viscosity or gel formation. it is used in salad dressings, sauces, desserts, baked goods, and beverages at 0.05–0.50%. |
| Uses | In foods, pharmaceuticals, and cosmetics as stabilizer and thickening agent. For rheology control in water-based systems. In oil and gas drilling and completion fluids. |
| Uses | xanthan gum (corn starch gum) serves as a texturizer, carrier agent, and gelling agent in cosmetic preparations. It also stabilizes and thickens formulations. This gum is produced through a fermentation of carbohydrate and Xanthomonas campestris. |
| Production Methods | Xanthan gum is a polysaccharide produced by a pure-culture aerobic fermentation of a carbohydrate with Xanthomonas campestris. The polysaccharide is then purified by recovery with propan-2-ol, dried, and milled. |
| Brand name | Rhodigel (Vanderbilt). |
| General Description | As xanthan is a polysaccharide used in many applications such as a food additive, enzyme substrate or rheology modifier, it is useful to have a xanthan standard with a clearly defined narrow molecular weight distribution. Xanthan is produced by fermentation from Xanthomonas campestris. |
| Pharmaceutical Applications | Xanthan gum is widely used in oral and topical pharmaceutical formulations, cosmetics, and foods as a suspending and stabilizing agent. It is also used as a thickening and emulsifying agent. It is nontoxic, compatible with most other pharmaceutical ingredients, and has good stability and viscosity properties over a wide pH and temperature range. Xanthan gum gels show pseudoplastic behavior, the shear thinning being directly proportional to the shear rate. The viscosity returns to normal immediately on release of shear stress. Xanthan gum has been used as a suspending agent for conventional, dry and sustained-release suspensions. When xanthan gum is mixed with certain inorganic suspending agents, such as magnesium aluminum silicate, or organic gums, synergistic rheological effects occur. In general, mixtures of xanthan gum and magnesium aluminum silicate in ratios between 1 : 2 and 1 : 9 produce the optimum properties. Similarly, optimum synergistic effects are obtained with xanthan gum : guar gum ratios between 3 : 7 and 1 : 9. Although primarily used as a suspending agent, xanthan gum has also been used to prepare sustained-release matrix tablets. Controlled-release tablets of diltiazem hydrochloride prepared using xanthan gum have been reported to sustain the drug release in a predictable manner, and the drug release profiles of these tablets were not affected by pH and agitation rate. Xanthan gum has also been used to produce directly compressed matrices that display a high degree of swelling due to water uptake, and a small amount of erosion due to polymer relaxation. It has also been used in combination with chitosan, guar gum, galactomannan, and sodium alginate to prepare sustained-release matrix tablets. Xanthan gum has been used as a binder, and in combination with Konjac glucomannan is used as an excipient for controlled colonic drug delivery. Xanthan gum with boswellia (3 : 1) and guar gum (10 : 20) have shown the best release profiles for the colon-specific compression coated systems of 5- fluorouracil for the treatment of colorectal cancer. Xanthan gum has also been used with guar gum for the development of a floating drug delivery system.It has also has derivatized to sodium carboxymethyl xanthan gum and crosslinked with aluminum ions to prepare microparticles, as a carrier for protein delivery. Xanthan gum has been incorporated in an ophthalmic liquid dosage form, which interacts with mucin, thereby helping in the prolonged retention of the dosage form in the precorneal area. When added to liquid ophthalmics, xanthan gum delays the release of active substances, increasing the therapeutic activity of the pharmaceutical formulations. Xanthan gum can be used to increase the bioadhesive strength in vaginal formulations. Xanthan gum alone or with carbopol 974P has been used as a mucoadhesive controlled-release excipient for buccal drug delivery. Modified xanthan films have been used as a matrix system for transdermal delivery of atenolol. Xanthan gum has also been used as a gelling agent for topical formulations incorporating solid lipid nanoparticles of vitamin A or microemulsion of ibuprofen. A combined polymer system consisting of xanthan gum, carboxy methylcellulose and a polyvinyl pyrolidone backboned polymer has been used for relieving the symptoms of xerostomia. Xanthan gum can also be used as an excipient for spray-drying and freeze-drying processes for better results. It has been successfully used alone or in combination with agar for microbial culture media. Xanthan gum is also used as a hydrocolloid in the food industry, and in cosmetics it has been used as a thickening agent in shampoo. Polyphosphate with xanthum gum in soft drinks is suggested to be effective at reducing erosion of enamel |
| Safety Profile | When heated to decomposition it emits acrid smoke and irritating fumes. |
| Safety | Xanthan gum is widely used in oral and topical pharmaceutical formulations, cosmetics, and food products, and is generally regarded as nontoxic and nonirritant at the levels employed as a pharmaceutical excipient. The estimated acceptable daily intake for xanthan gum has been set by the WHO at up to 10 mg/kg body-weight. No eye or skin irritation has been observed in rabbits and no skin allergy has been observed in guinea pigs following skin exposure. No adverse effects were observed in long term feeding studies with rats (up to 1000 mg/kg/day) and dogs (up to 1000 mg/kg/day). No adverse effects were observed in a three-generation reproduction study with rats (up to 500 mg/kg/day). LD50 (dog, oral): >20 g/kg LD50 (rat, oral): >45 g/kg LD50 (mouse, oral): >1 g/kg LD50 (mouse, IP): >50 mg/kg LD50 (mouse, IV): 100–250 mg/kg |
| storage | Xanthan gum is a stable material. Aqueous solutions are stable over a wide pH range (pH 3–12), although they demonstrate maximum stability at pH 4–10 and temperatures of 10–60°C. Xanthan gum solutions of less than 1% w/v concentration may be adversely affected by higher than ambient temperatures: for example, viscosity is reduced. Xanthan gum provides the same thickening, stabilizing, and suspending properties during long-term storage at elevated temperatures as it does at ambient conditions. In addition, it ensures excellent freeze–thaw stability. Solutions are also stable in the presence of enzymes, salts, acids, and bases. Vanzan NF-ST is especially designed for use in systems containing high salt concentrations as it dissolves directly in salt solutions, and its viscosity is relatively unaffected by high salt levels as compared with general purpose grades. The bulk material should be stored in a well-closed container in a cool, dry place. |
| Incompatibilities | Xanthan gum is an anionic material and is not usually compatible with cationic surfactants, polymers, or preservatives, as precipitation occurs. Anionic and amphoteric surfactants at concentrations above 15% w/v cause precipitation of xanthan gum from a solution. Under highly alkaline conditions, polyvalent metal ions such as calcium cause gelation or precipitation; this may be inhibited by the addition of a glucoheptonate sequestrant. The presence of low levels of borates (<300 ppm) can also cause gelation. This may be avoided by increasing the boron ion concentration or by lowering the pH of a formulation to less than pH 5. The addition of ethylene glycol, sorbitol, or mannitol may also prevent this gelation. Xanthan gum is compatible with most synthetic and natural viscosity-increasing agents, many strong mineral acids, and up to 30% inorganic salts. If it is to be combined with cellulose derivatives, then xanthan gum free of cellulase should be used to prevent depolymerization of the cellulose derivative. Xanthan gum solutions are stable in the presence of up to 60% water-miscible organic solvents such as acetone, methanol, ethanol, or propan-2- ol. However, above this concentration precipitation or gelation occurs. The viscosity of xanthan gum solutions is considerably increased, or gelation occurs, in the presence of some materials such as ceratonia, guar gum, and magnesium aluminum silicate. This effect is most pronounced in deionized water and is reduced by the presence of salt. This interaction may be desirable in some instances and can be exploited to reduce the amount of xanthan gum used in a formulation. Xanthan gum is incompatible with oxidizing agents, some tablet film-coatings, carboxymethylcellulose sodium, dried aluminum hydroxide gel, and some active ingredients such as amitriptyline, tamoxifen, and verapamil. |
| Regulatory Status | GRAS listed. Accepted for use as a food additive in Europe. Included in the FDA Inactive Ingredients Database (oral solutions, suspensions, and tablets; rectal and topical preparations). Included in nonparenteral medicines licensed in the UK. Included in the Canadian List of Acceptable Non-medicinal Ingredients. |
Q&A
What is the payment terms?
What is the payment terms?
T/T, L/C, D/P, etc.
Which kind of package do you have?
Which kind of package do you have?
carton, Iron drums, IBC Drums, Flexitank, ISO TANK and bags
etc.
How can I get samples?
How can I get samples?
We provide you samples for free, but you have to pay for the
courier charges.
Can you give me a discount price?
Can you give me a discount price?
Yes. It depends on your qty.
- Escolher uma seleção resulta numa atualização de página completa.
O seu carrinho
O seu carrinho está vazio
Tem uma conta?
Inicie sessão para finalizar a compra mais rápido.

